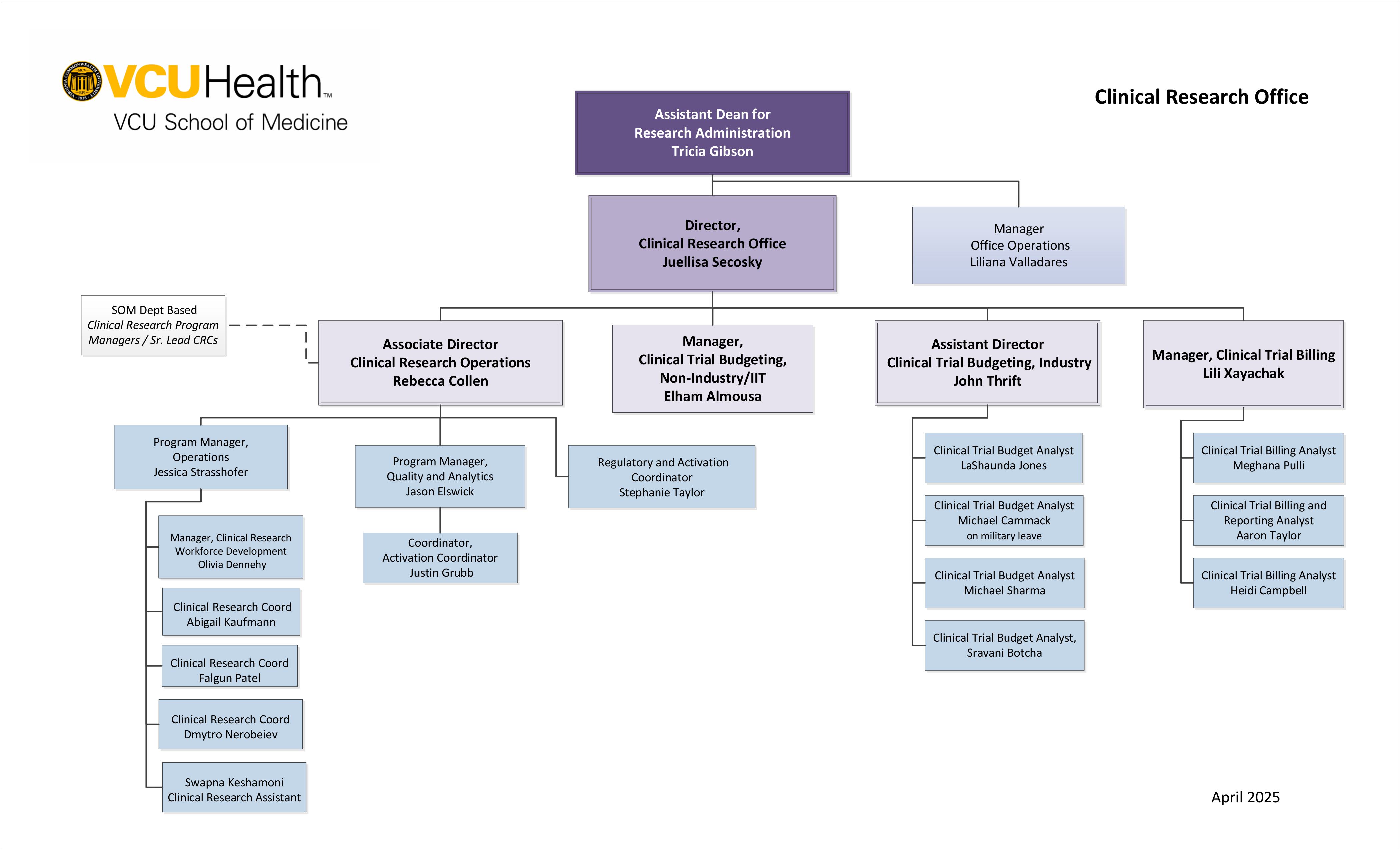
What is SOMCRO?
The School of Medicine Clinical Research Office (SOMCRO) consists of Budgets, Billing and Operations. SOMCRO works directly in collaboration with School of Medicine Research Administration to help investigators activate and manage clinical research projects in the School of Medicine.
What are SOMCRO's Goals?
- Supporting efficient study activation; directly overseeing administrative study activation in OnCore, coverage analysis, and budget (Budgets)
- Managing a shared/pooled resource of Clinical Research Coordinators (CRCs) supporting SOM Investigators (Operations)
- Maintaining CRC training programs for onboarding and continuous educational needs (Operations)
- Managing clinical research revenue recovery and clinical research billing utilizing the OnCore Financial Console (Billing)
- Developing and maintaining reporting and analytics to support data driven decision making at school and department levels for resource allocation, staffing, and program management (Data Analytics & Quality Assurance)
- Ongoing process improvement/efficiency to allow for continued growth of clinical research within the School of Medicine
Click to view as a PDF.
SOMCRO Units
The Budgets team serves as the point of contact for both new study activation and amendments for on-going studies, providing coverage analysis and budget development for faculty investigators and departments/divisions within the School of Medicine. The Budgets team also serves as a liaison to other areas/groups involved in study activation and study amendments.
Personnel
- John Thrift, Assistant Director Clinical Trial Budgeting (Industry)
- LaShaunda Jones, Clinical Trial Budget Analyst
- Michael Sharma, Clinical Trial Budget Analyst
- Sravani Botcha, Clinical Trial Budget Analyst
- Rebecca McDermott, Clinical Trial Budget Analyst
- Elham Almousa, Manager Clinical Trial Budgeting (non-industry/IIT)
Budget goals
- Be a trusted partner of SOM departments and divisions, faculty investigators and the study teams in support of their research goals and programmatic priorities.
- Ease the administrative burden in study activation by directly performing key aspects of study start-up.
- Be communicative and responsive to study teams regarding study status and departmental updates.
- Continually refine and share processes throughout the SOM related to clinical trial start-up, activation and ongoing management.
- Participate in the training of research personnel throughout the SOM, including being a resource for Good Clinical Practices (GCP) training as well as local process and best practice orientation.
Budget practices
- Administrative Startup Fees for Industry-Sponsored Clinical Trials — OVPRI Compliance Notice 17-006
- Startup Process Explanation
Contact information
For any questions related to new study activation, amendments, or to learn more about budgeting, please contact SOMCT@vcuhealth.org.
Personnel
- Travis Brown, Assistant Director Clinical Trial Billing & Analytics
- Aaron Taylor, Clinical Trial Billing and Reporting Analyst
- Meghana Pulli, Clinical Trial Billing Analyst
- Heidi Campbell, Clinical Trial Billing Analyst
Billing responsibilities
The Billing team oversees a number of key responsibilities for the Clinical Research Office, including:
- Invoicing for studies: A monthly review of currently active studies to ensure all transactions are properly accounted for
- Accounts payable (AP) management: Reviewing all incoming bills from service ancillaries to ensure efficiency
- Accounts Receivable (AR) operations: Maintaining a precise record of financial transactions for studies by reconciling and reviewing incoming study payments
- Reporting and financial oversight: Conducting thorough reviews for new studies, managing the closure of completed studies, and generating comprehensive reports to provide detailed insights into financial aspects
Helpful tips
- Please provide a direct Sponsor email contact for billing purposes to ensure timely processing.
- When sending payment, please provide the payment details/backups so the itemized list may be properly reconciled.
- Payment details should be sent to both SOMCRO Billing as well as VCU’s Grants & Contract Accounting office.
Contact information
For invoicing, accounts payable/receivable, financial oversight or to learn more about billing, please contact ctfinance@vcuhealth.org.
Personnel
- Rebecca Collen, Associate Director Clinical Research Operations
- Jessica Strasshofer, Program Manager Operations
- Olivia Dennehy, Manager Clinical Research Workforce Development
- Falgun Patel, Clinical Research Coordinator
- Dmytro Nerobeiev, Clinical Research Coordinator
- Stephanie Taylor, Regulatory and Activation Coordinator
- Jason Elswick, Program Manager Quality and Analytics
- Justin Grubb, Activation Coordinator
- Swapna Keshamoni, Clinical Research Assistant
- Natalie Nerobeieva, Clinical Research Coordinator
- Nicholas Griffin, Clinical Research Coordinator
Operations responsibilities
The Operations team provides reliable and compliant clinical research coordination services to investigators in VCU SOM who need additional support, supporting investigators and research teams with the planning, implementation, and management of clinical trials.
Available to any division in need of its services, the Operations team supports any study procedure including initial regulatory startup and ongoing management, managing full protocols, and devoting effort toward trials when need arises.
Contact information
For information on coordinator services for a new or current project, or to learn more about the Operations team, please contact SOMCRO@vcuhealth.org or use the Coordination Request Form.
Systems, Links and Resources
University Systems
Create funding proposals, submit documents for review and manage your proposals and awards
A central system to manage all studies requiring IRB review
The enterprise-wide clinical research management system for VCU and VCU Health
PROC review and approval is required for any study that will utilize VCU Health resources
21 CFR part 11 compliant electronic regulatory system designed for clinical trials
VCU's electronic signature system for legally-binding agreements and contracts
University Resources
VCU's central office for administrative award-related matters
Provides guidance and resources for research involving human participants
Provides training, support and collaboration opportunities to advance translational research
Searchable list of research-related policies and guidelines
Regulatory requirements, training, and contact information for all types of research
Required for all studies using VCU Health patients or facilities (following PROC approval)
School of Medicine Resources
CROC offers specialized clinical research coordinator services
Additional information about SOMCRO available on the Intranet for VCU personnel
Additional information about the CRC available on the Intranet for VCU personnel
Additional information about the VCUH OCR available on the Intranet for VCU personnel
Click to view as a PDF.
Shared Processes
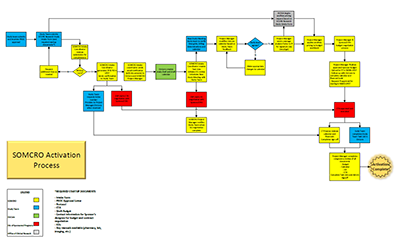
New study activation process
Looking to get a new study up and running? SOMCRO provides support and guidance throughout the activation process.
For step-by-step instructions, as well as easy access to documents and other resources, visit our Operations team's Clinical Research Activation Process page on Sharepoint.
Contacts
If you have questions related to:
- Invoicing, accounts payable/receivable, financial oversight:
CTFinance@vcuhealth.org - Coordinator services for a new or current project:
SOMCRO@vcuhealth.org - New study activation or amendments to current studies:
SOMCT@vcuhealth.org
